 There’s nothing wrong with that, but I think it gets a little daunting when it comes to having to keep track of multiple numbers of atoms (like 5 or more) and it will prolong the process unnecessarily. 
Most of the students I have worked with only knew Method 1 from their high school science/chemistry class. My quick google search seems to point towards Method 1 (by inspection or trial-error-method) and Method 2 (writing down atom counts) being the most popular methods when it comes to balancing equations. It does not indicate that the observed value is somehow better than expected, since the best possible outcome for percentage error is that the observed and true values are equal, resulting in a percentage error of 0.Last week I posted on the three methods to balance chemical equations. Thus, in the context of an experiment, a negative percentage error just means that the measured value is smaller than expected. If the observed value is larger than the true value, the percentage error will be positive. 
For example, given an observed value of 7, a true value of 9, and allowing for a negative percentage, the percentage error is: v observed – v trueĪ negative percentage error simply means that the observed value is smaller than the true value. 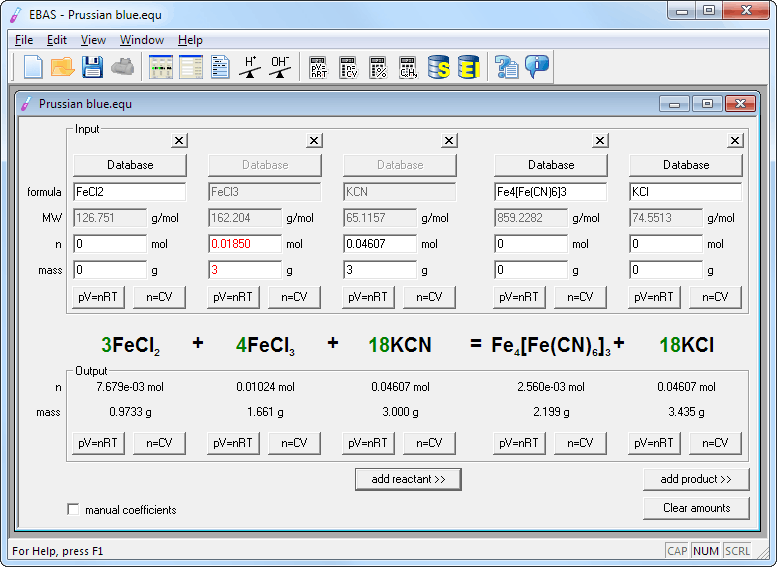
This occurs if we do not take the absolute value of the error, the observed value is smaller than the true value, and the true value is positive. However, it is possible to have a negative percentage error. In most cases, only the error is important, and not the direction of the error. Negative percentage errorīased on the formula above, when the true value is positive, percentage error is always positive due to the absolute value. Please refer to the standard deviation calculator for further details. True values are often unknown, and under these situations, standard deviation is one way to represent the error. The equations above are based on the assumption that true values are known. Absolute error = |V observed – V true|įor example, if the observed value is 56.891 and the true value is 62.327, the percentage error is: |56.891 – 62.327| Refer to the equations below for clarification. The absolute error is then divided by the true value, resulting in the relative error, which is multiplied by 100 to obtain the percentage error. The computation of percentage error involves the use of the absolute error, which is simply the difference between the observed and the true value. 
If, for example, the measured value varies from the expected value by 90%, there is likely an error, or the method of measurement may not be accurate. In most cases, a small percentage error is desirable, while a large percentage error may indicate an error or that an experiment or measurement technique may need to be re-evaluated. A small percentage error means that the observed and true value are close while a large percentage error indicates that the observed and true value vary greatly. Calculating the percentage error provides a means to quantify the degree by which a measured value varies relative to the true value. Error can arise due to many different reasons that are often related to human error, but can also be due to estimations and limitations of devices used in measurement. When measuring data, whether it be the density of some material, standard acceleration due to gravity of a falling object, or something else entirely, the measured value often varies from the true value. known values as well as to assess whether the measurements taken are valid. It is typically used to compare measured vs. Percentage error is a measurement of the discrepancy between an observed (measured) and a true (expected, accepted, known etc.) value. Related Percentage Calculator | Scientific Calculator | Statistics Calculator Percentage Error 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
